IGCSE Physics | Core & Supplement
1. What is Density? #
The equation for density is:
$$\rho = \frac{m}{V}$$- $\rho$ (rho) = density ($\text{g/cm}^3$ or $\text{kg/m}^3$)
- $m$ = mass ($\text{g}$ or $\text{kg}$)
- $V$ = volume ($\text{cm}^3$ or $\text{m}^3$)
You can rearrange this formula to find mass or volume:
$$m = \rho \times V \qquad\qquad V = \frac{m}{\rho}$$A piece of quartz has mass 200 g and density $2.5\ \text{g/cm}^3$. Find its volume.
- Formula: $$V = \frac{m}{\rho}$$
- Given: $m = 200\ \text{g}$, $\rho = 2.5\ \text{g/cm}^3$
- Express 2.5 as a fraction, then apply KCF (Keep, Change, Flip): $$V = 200 \div \frac{5}{2} = \frac{200}{1} \times \frac{2}{5}$$
- Cross-cancel — top-left 200 and bottom-right 5 share a factor of 5: $$V = \frac{\cancelto{40}{200}}{1} \times \frac{2}{\cancelto{1}{5}} = \frac{40 \times 2}{1 \times 1}$$
- Answer: $$V = 80\ \text{cm}^3$$
2. Measuring Density #
2a. Density of a Liquid #
- Place an empty measuring cylinder on a balance. Record its mass as $m_1$.
- Pour the liquid into the cylinder. Read the volume $V$ from the scale.
- Record the new mass of the cylinder + liquid as $m_2$.
- Mass of liquid: $m = m_2 – m_1$
- Calculate density: $\rho = \dfrac{m}{V}$
Empty cylinder mass = 80 g. Cylinder + liquid mass = 160 g. Volume of liquid = 50 cm³.
- Formula: $$\rho = \frac{m}{V}$$
- Given: $m = 160 – 80 = 80\ \text{g}$, $V = 50\ \text{cm}^3$
- Substitute: $$\rho = \frac{80}{50}$$
- Answer: $$\rho = 1.6\ \text{g/cm}^3$$
2b. Density of a Regularly Shaped Solid #
A regularly shaped solid has a shape with a known volume formula — for example a cube, rectangular block, or cylinder.
- Use a ruler (or vernier calipers) to measure the dimensions.
- Calculate the volume using the correct formula.
For a rectangular block: $V = \text{length} \times \text{width} \times \text{height}$ - Use a balance to measure the mass.
- Calculate density: $\rho = \dfrac{m}{V}$
A metal cube has sides of 4 cm each. Its mass is 512 g.
- Formula: $$\rho = \frac{m}{V}$$
- Find volume: $$V = 4 \times 4 \times 4 = 64\ \text{cm}^3$$
- Given: $m = 512\ \text{g}$, $V = 64\ \text{cm}^3$
- Substitute: $$\rho = \frac{512}{64}$$
- Answer: $$\rho = 8\ \text{g/cm}^3$$
2c. Density of an Irregularly Shaped Solid (Volume by Displacement) #
An irregularly shaped solid (like a rock) has no simple formula for volume. We find its volume by measuring how much water it pushes aside — this is called volume by displacement.
- Use a balance to measure the mass of the solid.
- Fill a measuring cylinder with water. Record the initial volume $V_1$.
- Carefully lower the solid into the water (make sure it is fully submerged). Record the new volume $V_2$.
- Volume of solid: $V = V_2 – V_1$
- Calculate density: $\rho = \dfrac{m}{V}$
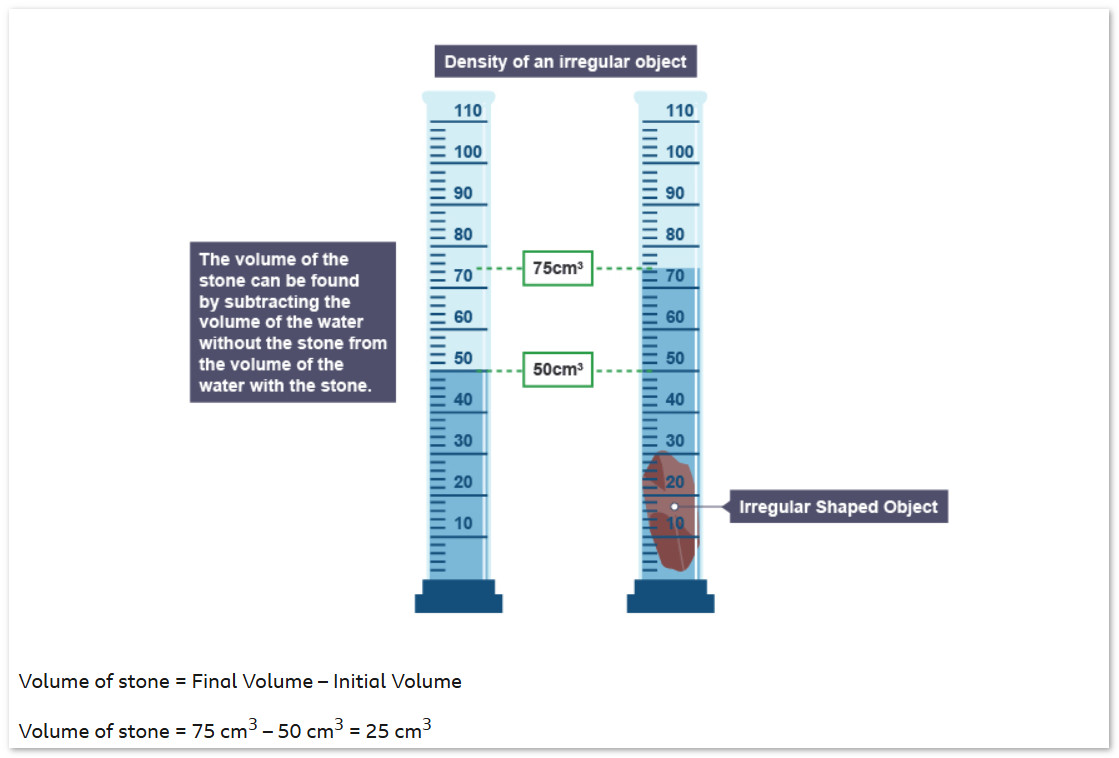
A rock has mass 180 g. Water level before = 40 cm³. Water level after submerging the rock = 70 cm³.
- Formula: $$\rho = \frac{m}{V}$$
- Find volume: $$V = 70 – 40 = 30\ \text{cm}^3$$
- Given: $m = 180\ \text{g}$, $V = 30\ \text{cm}^3$
- Substitute: $$\rho = \frac{180}{30}$$
- Answer: $$\rho = 6\ \text{g/cm}^3$$
3. Floating and Sinking #
You can use density to predict whether an object will float or sink in a liquid.
- Object density less than liquid density → object floats
- Object density greater than liquid density → object sinks
- Object density equal to liquid density → object stays suspended (does not float or sink)
Water density = $1.0\ \text{g/cm}^3$
| Object | Density (g/cm³) | Comparison | Result |
|---|---|---|---|
| Wood block | 0.6 | $0.6 < 1.0$ | Floats |
| Ice | 0.92 | $0.92 < 1.0$ | Floats |
| Steel bolt | 7.8 | $7.8 > 1.0$ | Sinks |
4. Liquids Floating on Other Liquids Supplement #
When two liquids that do not mix are placed together, the less dense liquid rises to the top and the more dense liquid sinks to the bottom.
- Lower density liquid → floats on top
- Higher density liquid → sinks to the bottom
Vegetable oil has density $0.9\ \text{g/cm}^3$. Water has density $1.0\ \text{g/cm}^3$. They do not mix.
Since $0.9 < 1.0$, the oil has a lower density than water.
Conclusion: The oil floats on top of the water.
Syllabus Reference — 1.4 Density #
Core
- Define density as mass per unit volume; recall and use the equation $\rho = \dfrac{m}{V}$
- Describe how to determine the density of a liquid, of a regularly shaped solid and of an irregularly shaped solid which sinks in a liquid (volume by displacement), including appropriate calculations
- Determine whether an object floats based on density data
Supplement
- Determine whether one liquid will float on another liquid based on density data given that the liquids do not mix




