Table of Contents
Introduction #
Chemical formulas are like a language that chemists use to describe molecules and compounds. They tell us what elements are present in a compound and how many atoms of each element we have. Understanding how to write and interpret chemical formulas is fundamental to chemistry.
Writing Chemical Formulas #
Basic Rules #
- Elements and Symbols
- Each element has a unique symbol (e.g., Na for Sodium, Cl for Chlorine)
- The first letter is always s Capital Letter
- If there’s a second letter, it’s lowercase
- Numbers in Formulas
- Numbers written just below each atom/compound, show how many atoms of each element
- If no number is written, it means there’s one atom
Examples from Questions #
Let’s look at some examples from Question 1:
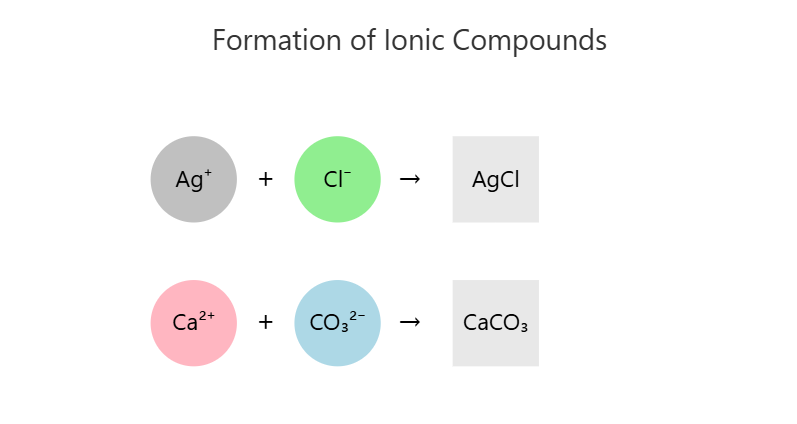
- Silver Chloride (AgCl)
- Made from Ag⁺ and Cl⁻
- One silver ion combines with one chloride ion
- Simple 1:1 ratio gives AgCl
- Calcium Carbonate (CaCO₃)
- Made from Ca²⁺ and CO₃²⁻
- One calcium ion combines with one carbonate ion
- Formula becomes CaCO₃