Table of Contents
Introduction #
Ions are atoms or groups of atoms that have gained or lost electrons, giving them an electrical charge. Understanding common ions is crucial for writing chemical formulas and predicting chemical reactions.
Positive Ions (Cations) #
- Group 1 Metals (Always +1)
- Lithium (Li⁺)
- Sodium (Na⁺)
- Potassium (K⁺)
- Rubidium (Rb⁺)
- Cesium (Cs⁺)
- Group 2 Metals (Always +2)
- Magnesium (Mg²⁺)
- Calcium (Ca²⁺)
- Strontium (Sr²⁺)
- Barium (Ba²⁺)
- Transition Metals (Variable Charges)
- Iron: Fe²⁺ (Iron II) or Fe³⁺ (Iron III)
- Copper: Cu⁺ (Copper I) or Cu²⁺ (Copper II)
- Zinc: Zn²⁺
- Silver: Ag⁺
- Gold: Au⁺ (Gold I) or Au³⁺ (Gold III)
Negative Ions (Anions) #
- Single Element Anions
- Fluoride (F⁻)
- Chloride (Cl⁻)
- Bromide (Br⁻)
- Iodide (I⁻)
- Oxide (O²⁻)
- Sulfide (S²⁻)
Polyatomic Ions #
These are groups of atoms that join together behave as if they are a single atom. They always carry a charge.
- Common Positive Polyatomic Ions
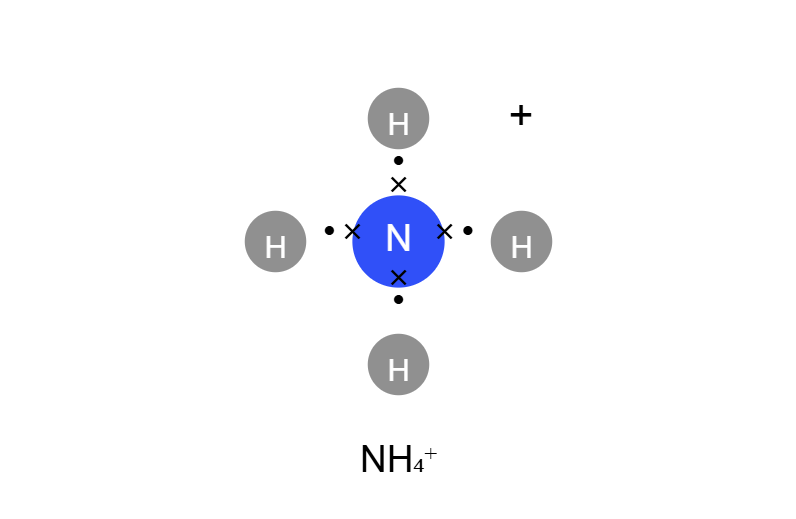
- Ammonium (NH₄⁺)
- Common Negative Polyatomic Ions
- Carbonate (CO₃²⁻)
- Bicarbonate (HCO₃⁻)
- Hydrogen sulfate (HSO₄⁻)
- Permanganate (MnO₄⁻)
- Nitrate (NO₃⁻)
- Nitrite (NO₂⁻)
- Sulfate (SO₄²⁻)
- Sulfite (SO₃²⁻)
- Phosphate (PO₄³⁻)
- Chlorite (ClO₂⁻)
Key Points to Remember #
- Charge Patterns
- Group 1 metals always form +1 ions
- Group 2 metals always form +2 ions
- Group 17 (halogens) always form -1 ions
- Transition metals can have multiple possible charges




